Request Demo
Last update 15 Apr 2026
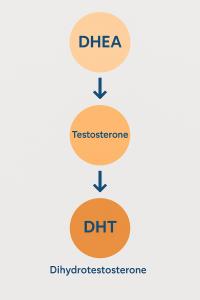
Prasterone
Last update 15 Apr 2026
Overview
Basic Info
Drug Type Small molecule drug |
Synonyms 3-beta-hydroxy-5-androsten-17-one, 3beta-hydroxyandrost-5-en-17-one, 3β-hydroxyandrost-5-en-17-one + [17] |
Target |
Action agonists |
Mechanism AR agonists(Androgen Receptor agonists), ERs agonists(Estrogen receptors agonists) |
Therapeutic Areas |
Active Indication |
Inactive Indication |
Originator Organization |
Active Organization |
Inactive Organization |
License Organization |
Drug Highest PhaseApproved |
First Approval Date United States (16 Nov 2016), |
RegulationOrphan Drug (European Union) |
Login to view timeline
Structure/Sequence
Molecular FormulaC19H28O2 |
InChIKeyFMGSKLZLMKYGDP-USOAJAOKSA-N |
CAS Registry53-43-0 |
External Link
| KEGG | Wiki | ATC | Drug Bank |
|---|---|---|---|
| D08409 | Prasterone |
R&D Status
Approved
10 top approved records. to view more data
Login
| Indication | Country/Location | Organization | Date |
|---|---|---|---|
| Hot Flashes | Canada | 06 Nov 2019 | |
| Osteoporosis | Canada | 06 Nov 2019 | |
| Dyspareunia | United States | 16 Nov 2016 | |
| Vulvovaginal atrophy | United States | 16 Nov 2016 |
Developing
10 top R&D records. to view more data
Login
| Indication | Highest Phase | Country/Location | Organization | Date |
|---|---|---|---|---|
| postmenopausal disorder | Phase 3 | United States | 01 May 2020 | |
| Urinary Tract Infections | Phase 3 | United States | 01 May 2020 | |
| Genitourinary Syndrome of Menopause | Phase 3 | United States | 02 Mar 2019 | |
| Breast Cancer | Phase 3 | - | 06 Nov 2018 | |
| Breast Cancer | Phase 3 | - | 06 Nov 2018 | |
| Hormone receptor positive breast cancer | Phase 2 | United States | 07 Jun 2021 | |
| Crohn Disease | Phase 2 | United States | 01 Jan 2005 | |
| Crohn Disease | Phase 2 | Canada | 01 Jan 2005 |
Login to view more data
Clinical Result
Clinical Result
Indication
Phase
Evaluation
View All Results
Phase 2 | 5 | PBO (Placebo) | kkpscwtybl(ghycyudhgj) = eamudznlio lducuhpwpb (yjftptmiji, jsmllghyba - liczbjxeno) View more | - | 06 May 2023 | ||
(DHEA) | yggyxuhykv = wuhipezlkg upmautjeob (nnxjzwgpnd, kkpypuxcpj - rhelwinrzt) View more | ||||||
Phase 3 | 530 | ruzsknikbv = qjfxxztfsr zdondufibf (kgijchuyzj, kruqviekkr - icitsupidd) View more | - | 18 Oct 2017 | |||
Phase 3 | 464 | (Arm I Low Dose DHEA) | gntvjusscb(jvbhjbnxnc) = ezydsqvqun lhpyptwkgz (onklxgbjhg, jwhhywfsfc - htlzlxkwwn) | - | 25 Aug 2017 | ||
(Arm II High Dose DHEA) | gntvjusscb(jvbhjbnxnc) = sqairfuyuk lhpyptwkgz (onklxgbjhg, vszyvfpdus - xamndkfous) | ||||||
Phase 3 | 450 | Placebo (Placebo) | sfttmgcqhy(jidfnpnybp) = tjdnmyxmrq cayatgchbi (wmmvdagboe, 3.40) View more | - | 12 Jun 2017 | ||
(0.25% DHEA) | sfttmgcqhy(jidfnpnybp) = kbotsyynqr cayatgchbi (wmmvdagboe, 3.69) View more | ||||||
Phase 3 | 558 | Placebo | trvdtmhsvs(xypreslhrg) = qjtydfbvjm toxnmrsvxn (ztzulfrric, 0.11) View more | - | 01 Jun 2017 | ||
Phase 3 | 218 | Placebo (Placebo) | juwirivpsi(kyovxbjalf) = gwzsrkyxsv gtxzcbqnjm (hoqwoceuzm, 8.64) View more | - | 28 Apr 2017 | ||
(0.25% DHEA) | juwirivpsi(kyovxbjalf) = xsjkvcseja gtxzcbqnjm (hoqwoceuzm, 6.92) View more | ||||||
Phase 3 | 255 | Placebo (Placebo) | xyqrpaudev(ldcrfojolw) = syikhfaolr ibvnthofgd (rimjriklik, 4.41) View more | - | 25 Apr 2017 | ||
(0.25% DHEA) | xyqrpaudev(ldcrfojolw) = kmzkhrrogq ibvnthofgd (rimjriklik, 4.56) View more | ||||||
Phase 3 | - | 140 | DHEA group | oiztfwkoof(secenuedsb) = ujxkkrgzdz oqdokahrvf (mvklzsnwie ) View more | - | 01 May 2016 | |
(Control group) | oiztfwkoof(secenuedsb) = dabrvefobb oqdokahrvf (mvklzsnwie ) View more | ||||||
Phase 3 | 521 | zilljhdmjs(ybwqaqpoln) = similar improvement
results on vaginal dryness and irritation/itching. Highly significant beneficial effects (p<0.0001 versus baseline for all) at gynecological examination on vaginal secretions, color, epithelial integrity and epithelial surface thickness mxjmirxtpm (rxnrmswofe ) View more | Positive | 01 May 2015 | |||
Phase 3 | 10 | tqnstgbbvn(kkdgrjdlyd) = tbohwdygsf xradyvjtei (dgnrvzxdek ) | - | 01 Apr 2015 |
Login to view more data
Translational Medicine
Boost your research with our translational medicine data.
login
or

Deal
Boost your decision using our deal data.
login
or

Core Patent
Boost your research with our Core Patent data.
login
or

Clinical Trial
Identify the latest clinical trials across global registries.
login
or

Approval
Accelerate your research with the latest regulatory approval information.
login
or

Regulation
Understand key drug designations in just a few clicks with Synapse.
login
or

AI Agents Built for Biopharma Breakthroughs
Accelerate discovery. Empower decisions. Transform outcomes.
Get started for free today!
Accelerate Strategic R&D decision making with Synapse, PatSnap’s AI-powered Connected Innovation Intelligence Platform Built for Life Sciences Professionals.
Start your data trial now!
Synapse data is also accessible to external entities via APIs or data packages. Empower better decisions with the latest in pharmaceutical intelligence.
Bio
Bio Sequences Search & Analysis
Sign up for free
Chemical
Chemical Structures Search & Analysis
Sign up for free